Authors: Ksenia Shinkar, Shapovalova O.E., Drozdov A. S.
Abstract
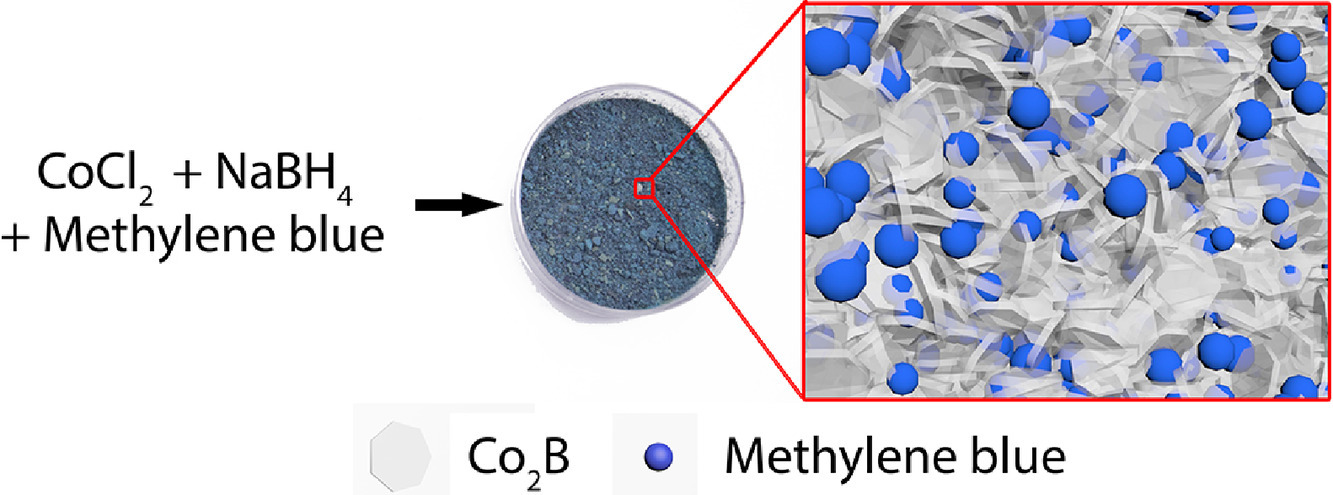
Here we describe the first example of organically doped cobalt borides materials by entrapment of model small compounds and proteins in the course of Co2+ ions reduction by sodium borohydride at room temperature. At these synthetic conditions, cobalt ions are reduced, and amorphous cobalt boride matrices are formed. The resulting materials showed superparamagnetic behavior with magnetization values up to 14 emu/g at 10 kOe. The formed mesoporous materials had developed structures; the estimated total surface areas equaled up to 48 m2/g with an average pore diameter of 2 nm. By carrying out the reaction in the presence of methylene blue as a model compound, composites were produced with up to 16.2% wt. mass fraction of the dopant. Mild synthetic conditions allowed to entrap biopolymers such as proteins or enzymes and preserve their structure at elevated temperatures. For instance, the entrapment of carbonic anhydrase enhanced its temperature of denaturation for 13 ∘C according to differential scanning calorimetry; the enzyme retained its catalytic activity at 80 ∘C, while free enzyme denaturated at 70 ∘C.
https://doi.org/10.1016/j.jtice.2020.04.011
Read Full: https://www.sciencedirect.com/science/article/abs/pii/S187610702030095X