Authors: Agafonov A.V., Afanasyev D.A., Gerasimova T.V., Krayev A.C., Kashirin M.A., Vinogradov V.V., Vinogradov A.V., Kessler V.G.
Abstract
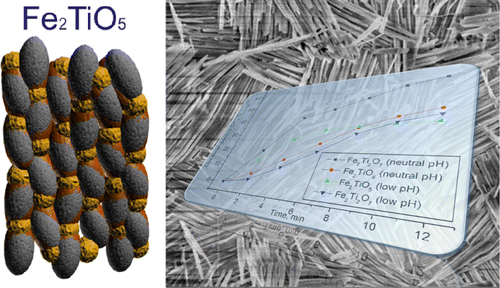
Colloidal synthesis of iron titanate-based nanocomposites exploiting the interaction of solutions of binary oxide nanoparticles, Fe2O3 and TiO2, was investigated with respect to the pH of the reaction medium and the conditions used for the synthesis of the reactants. It has been demonstrated that while the phase composition of the products is rather analogous, involving the formation of iron titanate phases on the grain boundaries of the binary oxide particles, the morphology of the resulting aggregates can be a matter of pH control. The self-assembly mechanisms are guided by the surface charge of the particles, offering nanorod regular colloid crystal structures of altering particles with opposite initial charges at neutral pH and globular aggregates with random distribution of uniformly charged particles at low pH as revealed by DLS and high-resolution TEM studies. The produced materials demonstrated enhanced photocatalytic activity compared to the iron titanates produced by conventional techniques. Magnetic characteristics have also been investigated disclosing the possibility of magnetic separation for the Fe2TiO5 material, making it an attractive candidate for application in the sustainable remediation of wastewaters.
DOI: 10.1021/acssuschemeng.6b00317
Read Full Here:
https://pubs.acs.org/doi/pdf/10.1021/acssuschemeng.6b00317