Authors: Krivoshapkin P.V., Ivanets A.I., Torlopov M.A., Mikhaylov V.I., Srivastava V., Sillanpää M., Prozorovich V.G., Kouznetsova T.F., Koshevaya E.D., Krivoshapkina E.F.
Abstract
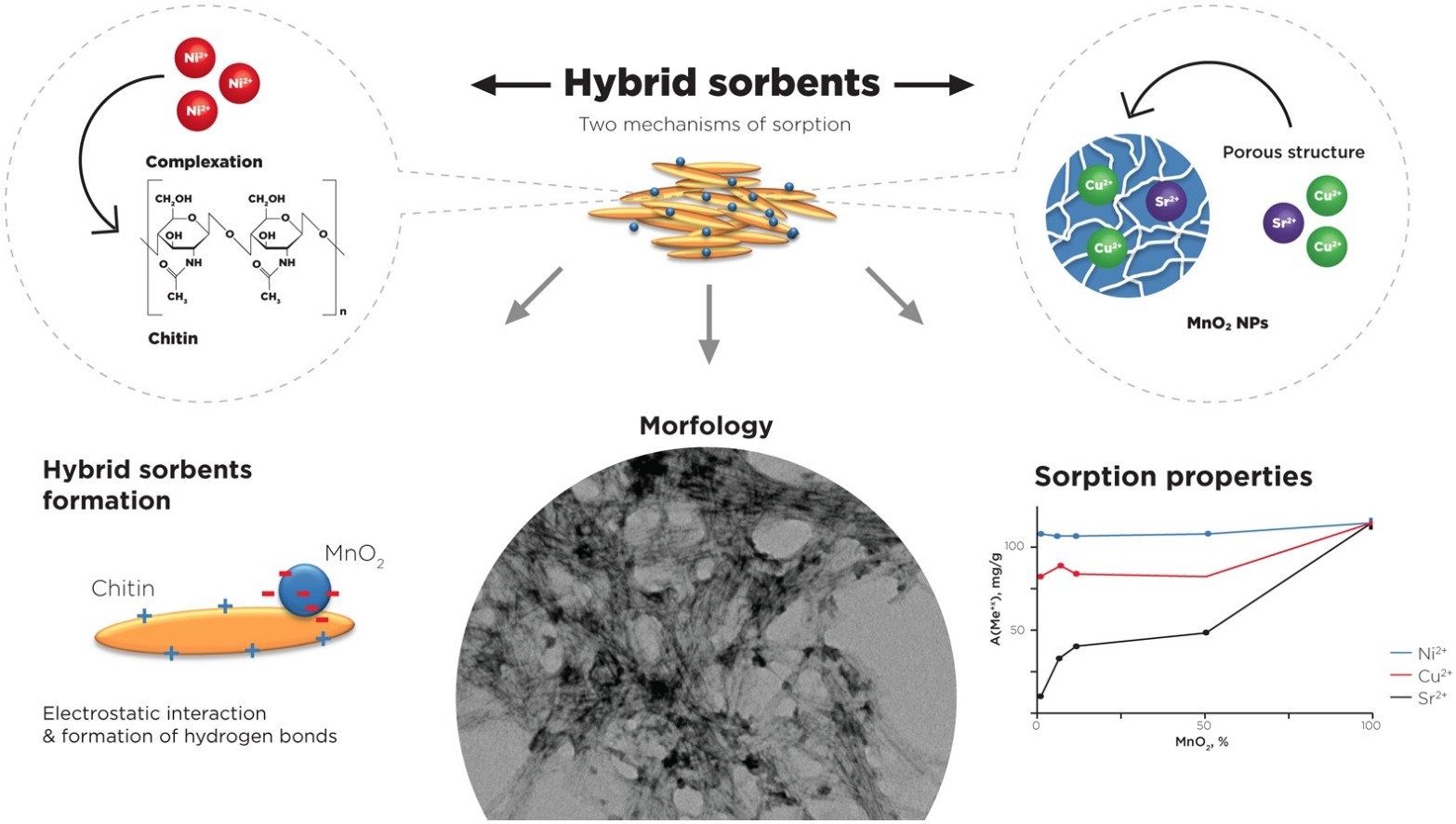
The article presents the results of the synthesis and research of a new biodegradable hybrid nanosized polymer-inorganic system possessing the double nature of heavy metal ions extraction from solutions. Firstly, the sorption of metal ions takes place by the ion-exchange properties of porous manganese oxide and, secondly, due to specific interaction and chelating with functional groups of polysaccharides in terms of nanochitin (ChNC). The synthesis is based on the colloid-chemical processes of interaction between polysaccharide and manganese oxide nanosized particles. Using the mathematical model of the Derjaguin, Landau, Verwey, and Overbeek (DLVO) theory, it has been shown that this interaction occurs due to the formation of hydrogen bonds and electrostatic interactions of oppositely charged particles. Hybrid sorbents were characterised by X-ray diffraction, Fourier transform infrared spectroscopy, Transmission electron and Atomic force microscopy. The highest value of sorption capacity for hybrid materials was obtained for Ni2+ extraction and reached 114.0 ± 1.1 mg/g. It was shown that biodegradation of hybrid sorbents increases with the concentration of manganese oxide nanoparticles and almost complete degradation of the organic part can be performed within 6 days. The obtained biodegradable sorbents were designed to address ecological problems connected with the pollution of natural waters by toxic metallic ions.
DOI: 10.1016/j.carbpol.2019.01.045
Read Full:
https://www.sciencedirect.com/science/article/pii/S014486171930058X#!