Authors: , Sergey N. Semenov
MDPI Life, 2019, 9 (2), 42
Abstract
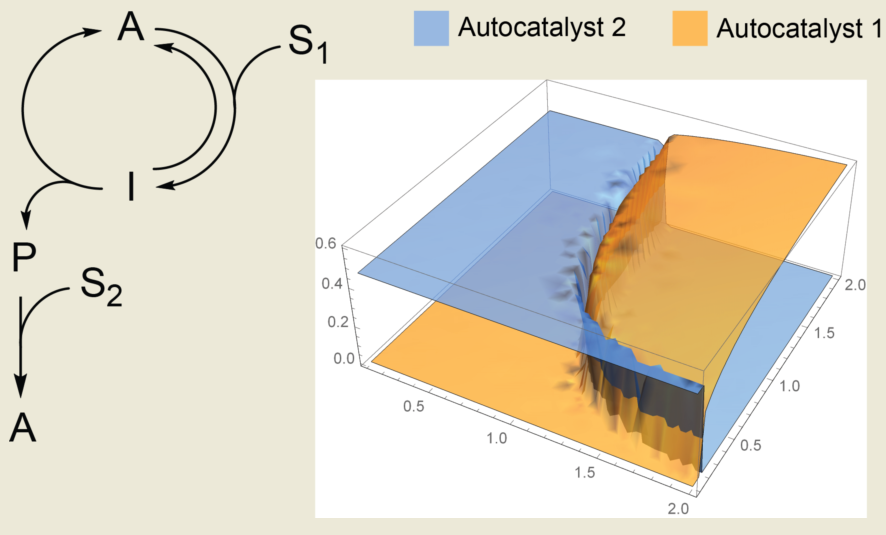
Network autocatalysis, which is autocatalysis whereby a catalyst is not directly produced in a catalytic cycle, is likely to be more common in chemistry than direct autocatalysis is. Nevertheless, the kinetics of autocatalytic networks often does not exactly follow simple quadratic or cubic rate laws and largely depends on the structure of the network. In this article, we analyzed one of the simplest and most chemically plausible autocatalytic networks where a catalytic cycle is coupled to an ancillary reaction that produces the catalyst. We analytically analyzed deviations in the kinetics of this network from its exponential growth and numerically studied the competition between two networks for common substrates. Our results showed that when quasi-steady-state approximation is applicable for at least one of the components, the deviation from the exponential growth is small. Numerical simulations showed that competition between networks results in the mutual exclusion of autocatalysts; however, the presence of a substantial noncatalytic conversion of substrates will create broad regions where autocatalysts can coexist. Thus, we should avoid the accumulation of intermediates and the noncatalytic conversion of the substrate when designing experimental systems that need autocatalysis as a source of positive feedback or as a source of evolutionary pressure.
DOI: 10.3390/life9020042
Read Full Here:
https://doi.org/10.3390/life9020042