Authors: Vasily I. Mikhaylov, Kryuchkova A.V., Petr A. Sitnikov, Liubov A. Koval, Nadezhda V. Zemskaya, Krivoshapkina E.F., Krivoshapkin P.V.
Abstract
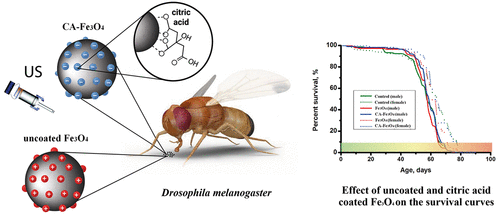
This paper presents sols of uncoated and citric acid-coated Fe3O4 nanoparticles obtained by a combination of coprecipitation and sonochemistry methods. A stable concentrated CA-Fe3O4 sol synthesized by a combination of coprecipitation with an inconvenient Fe2+/Fe3+ ratio, modification with citric acid and US treatment was obtained for the first time. A comparative analysis of the composition and morphology of nanoparticles was performed. The sols are oppositely charged and behave as a typical ferrofluid. The citric acid-modified sol is aggregatively stable over wider ranges of pH and electrolyte concentration, but it becomes less stable with the temperature increase. DLVO calculations showed that steric repulsion forces are a vital factor contributing to increased aggregative stability in a modified Fe3O4 sol. The experiments have revealed the magneto-optical effect in a modified Fe3O4 sol with an electrolyte concentration of 0.025–0.075 M caused by a high potential barrier and a deep secondary minimum in pairwise interaction curves. The “pK spectroscopy” mathematical model to describe the potentiometric curves of synthesized magnetite sols was used for the first time. According to potentiometric titration, the ions of the electrolyte practically do not contribute to formation of a surface charge in modified Fe3O4 with a change in pH due to blocking the magnetite surface by citric acid molecules. Drosophila melanogaster was used as a model to show that Fe3O4 in chronic exposure has a low toxic effect.
DOI: 10.1021/acs.langmuir.0c00605
Read Full: https://pubs.acs.org/doi/abs/10.1021/acs.langmuir.0c00605