Authors: Perovskiy I.A., Khramenkova E.V.,, Krivoshapkin P.V., Vinogradov A.V., Krivoshapkina E.F.
Abstract
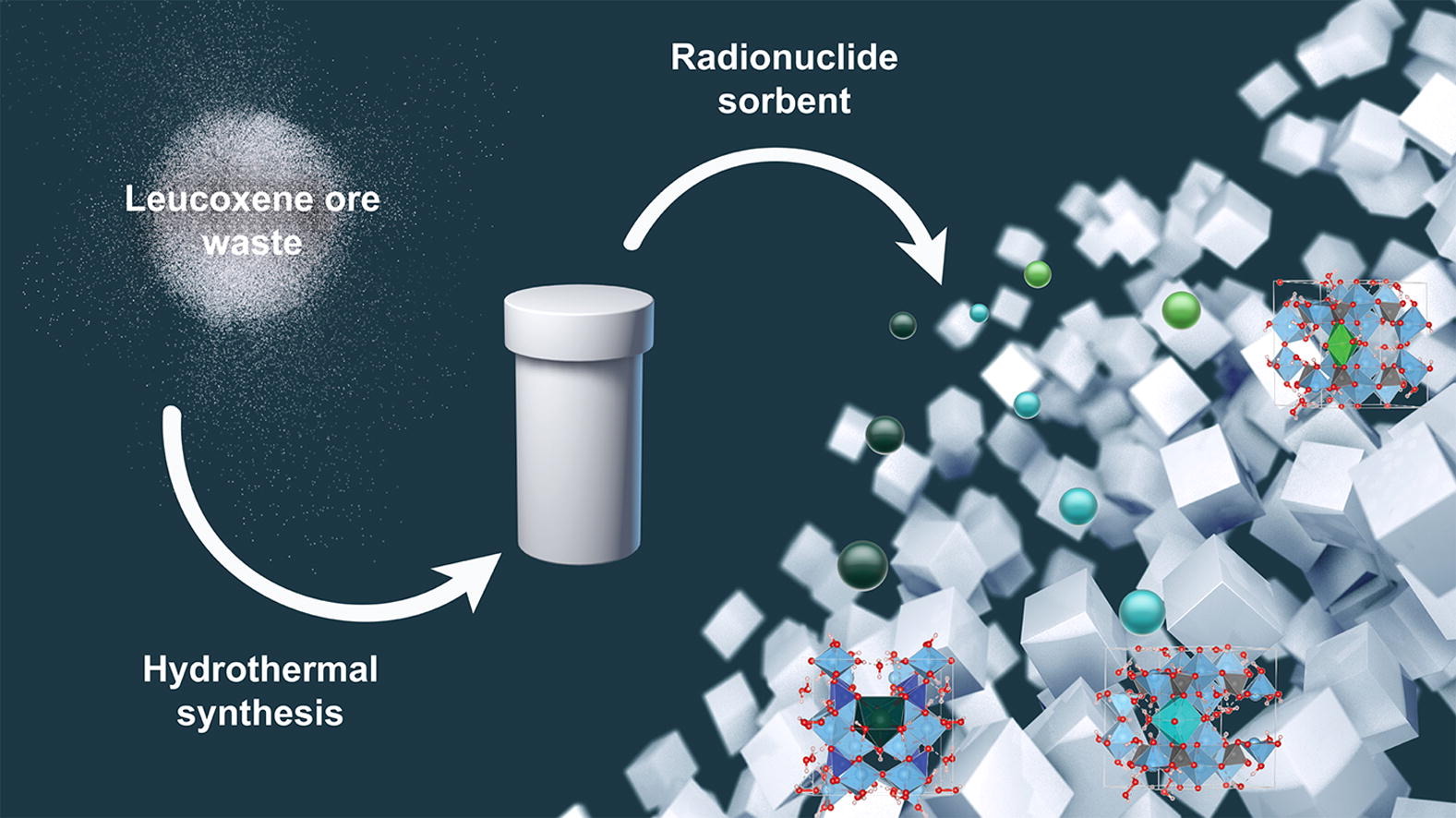
A highly selective adsorbent for multivalent cationic species based on a sitinakite-type titanosilicate was prepared from a leucoxene ore enrichment waste. The synthesized material was used as for the selective removal of alkali-earth strontium (II) and barium (II) cations as well as for the cationic species based on the natural isotopes of uranium, radium, and thorium from aqueous solutions. The influence of such parameters as the pH, the initial concentration of the ions, the presence of other electrolytes on the sorption parameters was investigated. The sorption capacity of the synthesized material at ambient conditions is 80 and 110 mg/g for Sr2+ and Ba2+, respectively, and it rises with increasing temperature. Furthermore, the material shows a high selectivity towards radionuclides of radium, uranium, and thorium. By using the current titanosilicate materials, the extracting degree of over 99% could be achieved when extracting these species from their respective standard aqueous solutions. The origin of the high adsorption selectivity for cationic complexes of thorium and uranium is rationalized based on periodic density functional theory calculations. The obtained results indicate that the described materials could be promising and inexpensive sorbents for the selective extraction of radioactive isotopes and particularly those of Sr, Ba and U, Th, Ra.
DOI: https://doi.org/10.1016/j.cej.2018.08.030
Read Full:
https://www.sciencedirect.com/science/article/pii/S1385894718314979