Авторы: ,
Abstract
Binary light‐up aptameric sensors (BLAS) consist of two RNA or DNA strands and a fluorogenic organic dye added as a buffer component. When associated, the two strands form a dye‐binding site, followed by an increase in fluorescence of the aptamer‐bound dye. This Minireview summarizes the state of the art in BLAS development and outlines perspectives and applications.
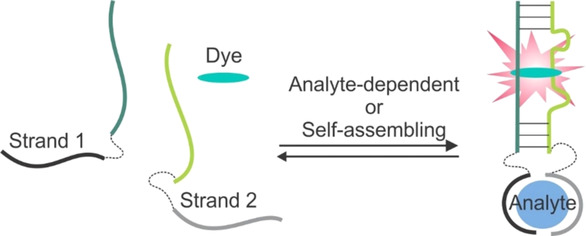
This review discusses the design and applications of binary (also known as split) light‐up aptameric sensors (BLAS). BLAS consist of two RNA or DNA strands and an fluorogenic organic dye added as a buffer component. When associate, the two strands form a dye‐binding site, followed by increase in fluorescence of the aptamer‐bound dye. The design is cost‐efficient since it uses short oligonucleotides and does not require conjugation of organic dyes with nucleic acids. In some applications, BLAS design is preferable over monolith sensors due to simpler assay optimization and improved selectivity. RNA‐based BLAS can be expressed in cells and used for intracellular monitoring of biological molecules. BLAS have been used as reporters of nucleic acid association events in RNA nanotechnology and nucleic acid‐based molecular computation. Other applications of BLAS include detection of nucleic acids, proteins and cancer cells, and potentially they can be tailored to report a broad range of biological analytes in a cost‐efficient, highly selective and easy‐to‐optimize format. This review summarizes the state of the art in BLAS development and outlines perspectives and applications
Keywords: fluorescent protein; label-free sensors; molecular beacon probe; in vivo imagine; RNA nanotechnology.
DOI: 10.1002/ange.201914919.
Read Full: https://onlinelibrary.wiley.